 
Something that surprises novices is that the (111) plane does not contain the body centered atom.It is the case that in a cubic 1:1 solid where one atom type is much larger than the other that the caesium chloride type lattice is obtained, it can be thought of as a combination of basketballs and golf balls packed in a cubic manner with the golf balls in the gaps between the basketballs. Notice that the (111) plane is not hexagonally closest packed as it is in the (111) plane of NaCl. The caesium chloride structure is composed of interlocking simple cubic lattices of anions and cations. 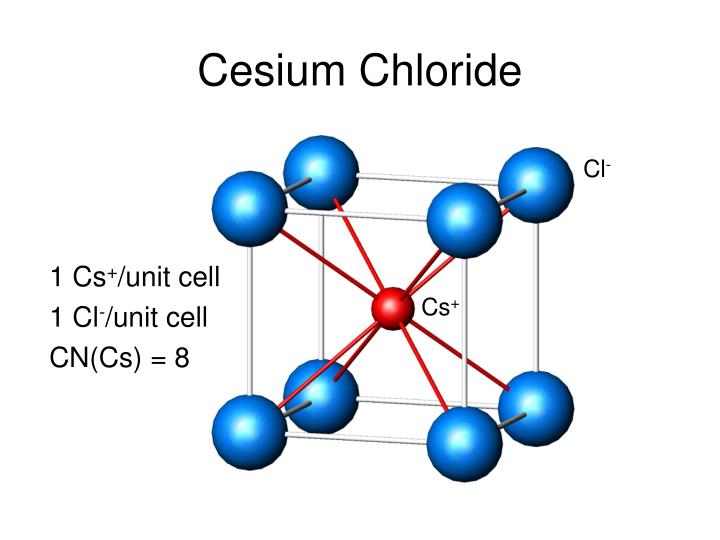
Shown below are two crystallographic planes in CsCl. The caesium chloride structure adopts a primitive cubic lattice with a two-atom basis, where both atoms have eightfold coordination. Each type of ion has a coordination number of 8. NH 4X, TlX (X = halide), CuZn (beta-brass), It can equivalently by described as cations at the corners of the cell with an anion at the centre. 
Structural Information CsCl Vital Statistics Note that each ion is 8-coordinate rather than 6-coordinate as in NaCl. As with NaCl, the 1:1 stoichiometry means that the cell will look the same regardless of whether we start with anions or cations on the corner. Tags: typical guest, charged Name: Cesium chloride, Caesium chloride Preferred Abbreviation: Cesium chloride IUPAC Name: cesium chloride CAS: CID. This is clearly not the case.ĬsCl can be thought of as two interpenetrating simple cubic arrays where the corner of one cell sits at the body center of the other. At first glance you might think that it is body-centered, but this would be true only if the atom at the body center was the same kind of atom as those on the corners of the cells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
